Fluid Mosaic Model
Scientists identified the plasma membrane in the 1890s, and its chemical components in 1915. The principal components they identified were lipids and proteins. In 1935, Hugh Davson and James Danielli proposed the plasma membrane's structure. This was the first model that others in the scientific community widely accepted. It was based on the plasma membrane's “railroad track” appearance in early electron micrographs. Davson and Danielli theorized that the plasma membrane's structure resembles a sandwich. They made the analogy of proteins to bread, and lipids to the filling. In the 1950s, advances in microscopy, notably transmission electron microscopy (TEM), allowed researchers to see that the plasma membrane's core consisted of a double, rather than a single, layer. In 1972, S.J. Singer and Garth L. Nicolson proposed a new model that provides microscopic observations and better explains plasma membrane function.
The explanation, the fluid mosaic model, has evolved somewhat over time, but it still best accounts for plasma membrane structure and function as we now understand them. The fluid mosaic model describes the plasma membrane structure as a mosaic of components—including phospholipids, cholesterol, proteins, and carbohydrates—that gives the membrane a fluid character. Plasma membranes range from 5 to 10 nm in thickness. For comparison, human red blood cells, visible via light microscopy, are approximately 8 µm wide, or approximately 1,000 times wider than a plasma membrane. The membrane does look a bit like a sandwich (Figure).

A plasma membrane's principal components are lipids (phospholipids and cholesterol), proteins, and carbohydrates attached to some of the lipids and proteins. A phospholipid is a molecule consisting of glycerol, two fatty acids, and a phosphate-linked head group. Cholesterol, another lipid comprised of four fused carbon rings, is situated alongside the phospholipids in the membrane's core. The protein, lipid, and carbohydrate proportions in the plasma membrane vary with cell type, but for a typical human cell, protein accounts for about 50 percent of the composition by mass, lipids (of all types) account for about 40 percent, and carbohydrates comprise the remaining 10 percent. However, protein and lipid concentration varies with different cell membranes. For example, myelin, an outgrowth of specialized cells' membrane that insulates the peripheral nerves' axons, contains only 18 percent protein and 76 percent lipid. The mitochondrial inner membrane contains 76 percent protein and only 24 percent lipid. The plasma membrane of human red blood cells is 30 percent lipid. Carbohydrates are present only on the plasma membrane's exterior surface and are attached to proteins, forming glycoproteins, or attached to lipids, forming glycolipids.
Phospholipids
The membrane's main fabric comprises amphiphilic, phospholipid molecules. The hydrophilic or “water-loving” areas of these molecules (which look like a collection of balls in an artist’s rendition of the model) (Figure) are in contact with the aqueous fluid both inside and outside the cell. Hydrophobic, or water-hating molecules, tend to be non-polar. They interact with other non-polar molecules in chemical reactions, but generally do not interact with polar molecules. When placed in water, hydrophobic molecules tend to form a ball or cluster. The phospholipids' hydrophilic regions form hydrogen bonds with water and other polar molecules on both the cell's exterior and interior. Thus, the membrane surfaces that face the cell's interior and exterior are hydrophilic. In contrast, the cell membrane's interior is hydrophobic and will not interact with water. Therefore, phospholipids form an excellent two-layer cell membrane that separates fluid within the cell from the fluid outside the cell.
A phospholipid molecule (Figure) consists of a three-carbon glycerol backbone with two fatty acid molecules attached to carbons 1 and 2, and a phosphate-containing group attached to the third carbon. This arrangement gives the overall molecule a head area (the phosphate-containing group), which has a polar character or negative charge, and a tail area (the fatty acids), which has no charge. The head can form hydrogen bonds, but the tail cannot. Scientists call a molecule with a positively or negatively charged area and an uncharged, or non-polar, area amphiphilic or “dual-loving.”
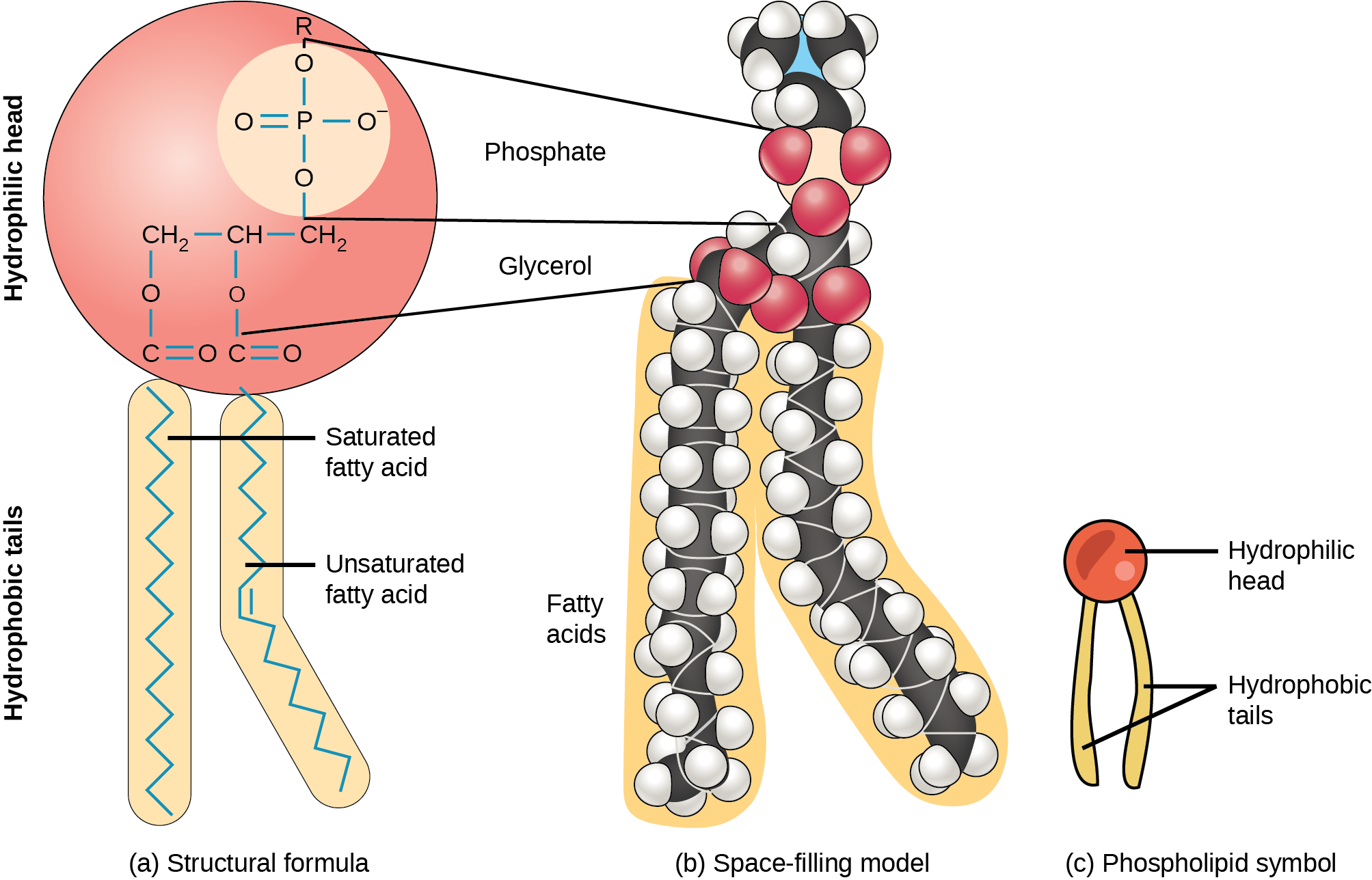
This characteristic is vital to the plasma membrane's structure because, in water, phospholipids arrange themselves with their hydrophobic tails facing each other and their hydrophilic heads facing out. In this way, they form a lipid bilayer—a double layered phospholipid barrier that separates the water and other materials on one side from the water and other materials on the other side. Phosopholipids heated in an aqueous solution usually spontaneously form small spheres or droplets (micelles or liposomes), with their hydrophilic heads forming the exterior and their hydrophobic tails on the inside (Figure).

Proteins
Proteins comprise the plasma membranes' second major component. Integral proteins, or integrins, as their name suggests, integrate completely into the membrane structure, and their hydrophobic membrane-spanning regions interact with the phospholipid bilayer's hydrophobic region (Figure). Single-pass integral membrane proteins usually have a hydrophobic transmembrane segment that consists of 20–25 amino acids. Some span only part of the membrane—associating with a single layer—while others stretch from one side to the other, and are exposed on either side. Up to 12 single protein segments comprise some complex proteins, which are extensively folded and embedded in the membrane (Figure). This protein type has a hydrophilic region or regions, and one or several mildly hydrophobic regions. This arrangement of protein regions orients the protein alongside the phospholipids, with the protein's hydrophobic region adjacent to the phosopholipids' tails and the protein's hydrophilic region or regions protruding from the membrane and in contact with the cytosol or extracellular fluid.

Peripheral proteins are on the membranes' exterior and interior surfaces, attached either to integral proteins or to phospholipids. Peripheral proteins, along with integral proteins, may serve as enzymes, as structural attachments for the cytoskeleton's fibers, or as part of the cell’s recognition sites. Scientists sometimes refer to these as “cell-specific” proteins. The body recognizes its own proteins and attacks foreign proteins associated with invasive pathogens.
Carbohydrates
Carbohydrates are the third major plasma membrane component. They are always on the cells' exterior surface and are bound either to proteins (forming glycoproteins) or to lipids (forming glycolipids) (Figure). These carbohydrate chains may consist of 2–60 monosaccharide units and can be either straight or branched. Along with peripheral proteins, carbohydrates form specialized sites on the cell surface that allow cells to recognize each other. These sites have unique patterns that allow for cell recognition, much the way that the facial features unique to each person allow individuals to recognize him or her. This recognition function is very important to cells, as it allows the immune system to differentiate between body cells (“self”) and foreign cells or tissues (“non-self”). Similar glycoprotein and glycolipid types are on the surfaces of viruses and may change frequently, preventing immune cells from recognizing and attacking them.
We collectively refer to these carbohydrates on the cell's exterior surface—the carbohydrate components of both glycoproteins and glycolipids—as the glycocalyx (meaning “sugar coating”). The glycocalyx is highly hydrophilic and attracts large amounts of water to the cell's surface. This aids in the cell's interaction with its watery environment and in the cell’s ability to obtain substances dissolved in the water. As we discussed above, the glycocalyx is also important for cell identification, self/non-self determination, and embryonic development, and is used in cell to cell attachments to form tissues.
Evolution Connection
How Viruses Infect Specific OrgansGlycoprotein and glycolipid patterns on the cells' surfaces give many viruses an opportunity for infection. HIV and hepatitis viruses infect only specific organs or cells in the human body. HIV is able to penetrate the plasma membranes of a subtype of lymphocytes called T-helper cells, as well as some monocytes and central nervous system cells. The hepatitis virus attacks liver cells.
These viruses are able to invade these cells, because the cells have binding sites on their surfaces that are specific to and compatible with certain viruses (Figure). Other recognition sites on the virus’s surface interact with the human immune system, prompting the body to produce antibodies. Antibodies are made in response to the antigens or proteins associated with invasive pathogens, or in response to foreign cells, such as might occur with an organ transplant. These same sites serve as places for antibodies to attach and either destroy or inhibit the virus' activity. Unfortunately, these recognition sites on HIV change at a rapid rate because of mutations, making an effective vaccine against the virus very difficult, as the virus evolves and adapts. A person infected with HIV will quickly develop different populations, or variants, of the virus that differences in these recognition sites distinguish. This rapid change of surface markers decreases the effectiveness of the person’s immune system in attacking the virus, because the antibodies will not recognize the surface patterns' new variations. In the case of HIV, the problem is compounded because the virus specifically infects and destroys cells involved in the immune response, further incapacitating the host.
